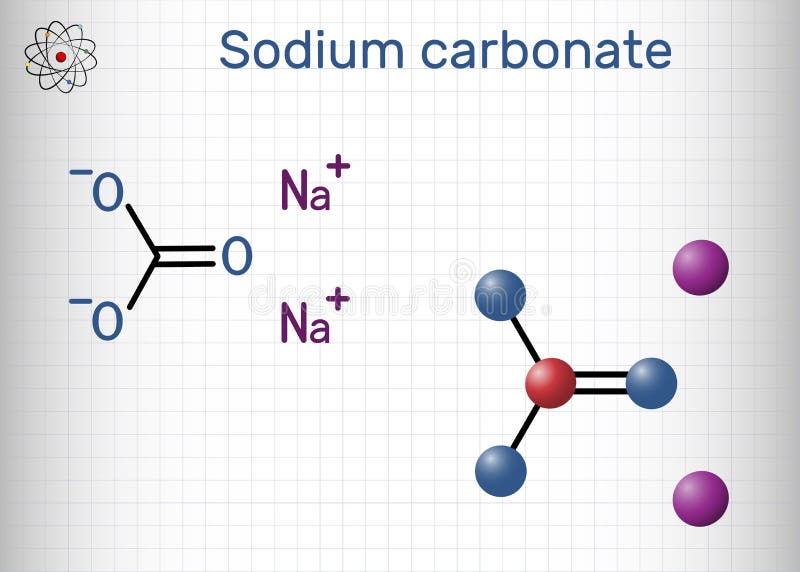
Sodium Carbonate (Washing Soda), Na2CO3⋅10H2O Sodium carbonate
By A Mystery Man Writer
Last updated 16 Jul 2024

Sodium Carbonate (Washing Soda), Na2CO3⋅10H2O
Sodium carbonate is generally prepared by Solvay Process. In this process, advantage is taken of the low solubility of sodium hydrogencarbonate whereby it gets precipitated in the reaction of sodium chloride with ammonium hydrogencarbonate. The latter is prepared by passing CO2 to a concentrated solution of sodium chloride saturated with ammonia, where ammonium carbonate followed by ammonium hydrogencarbonate are formed. The equations for the complete process may be written as :
2NH3+H2O+CO2→(NH4)2CO3(NH4)2CO3+H2O+CO2→2NH4HCO3NH4HCO3+NaCl→NH4Cl+NaHCO3
Sodium hydrogencarbonate crystal separates. These are heated to give sodium carbonate.
Video solution 1: Sodium Carbonate (Washing Soda), Na2CO3⋅10H2O Sodium carbonate is generally prepared by Solvay Process. In this process, advantage is taken of the low solubility of sodium hydrogencarbonate whereby it gets precipitated in the reaction of sodium chloride with ammonium hydrogencarbonate. The latter is prepared by passing CO2 to a concentrated solution of sodium chloride saturated with ammonia, where ammonium carbonate followed by ammonium hydrogencarbonate are formed. The equations for the complete process may be written as : 2NH3+H2O+CO2→(NH4)2CO3(NH4)2CO3+H2O+CO2→2NH4HCO3NH4HCO3+NaCl→NH4Cl+NaHCO3 Sodium hydrogencarbonate crystal separates. These are heated to give sodium carbonate.
Video solution 1: Sodium Carbonate (Washing Soda), Na2CO3⋅10H2O Sodium carbonate is generally prepared by Solvay Process. In this process, advantage is taken of the low solubility of sodium hydrogencarbonate whereby it gets precipitated in the reaction of sodium chloride with ammonium hydrogencarbonate. The latter is prepared by passing CO2 to a concentrated solution of sodium chloride saturated with ammonia, where ammonium carbonate followed by ammonium hydrogencarbonate are formed. The equations for the complete process may be written as : 2NH3+H2O+CO2→(NH4)2CO3(NH4)2CO3+H2O+CO2→2NH4HCO3NH4HCO3+NaCl→NH4Cl+NaHCO3 Sodium hydrogencarbonate crystal separates. These are heated to give sodium carbonate.

What Is Sodium Carbonate?

Sodium Carbonate Dense Soda Ash [Na2CO3] [CAS_497-19-8] NSF 99.6+%, Wh – Wintersun Chemical
Soda ash is used in a variety of industries and applications including:, • Glass Manufacturing, • Dry Powder Detergents (e.g., laundry, auto, dish,

Sodium Carbonate (Soda Ash)
Sodium carbonate also known as soda ash and washing soda is an alkaline product is used in production of detergents, water softening, and cooking

Sodium Carbonate, Soda Ash (NaCO3)
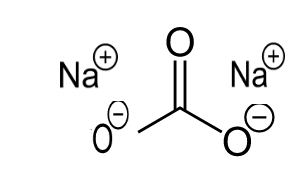
the story of washing soda na2co3 10h2o - Overview, Structure, Properties & Uses

Washing soda has the formula Na_(2) CO_(2).10H_(2)O. What is mass of anhydrous sodium carbonate

10 lb Pure Sodium Carbonate Dense Soda Ash Na2CO3 pH Adjust Chemical Spa Pool Cleaning Dishwasher : Health & Household
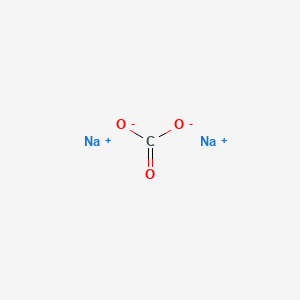
Sodium Carbonate, Na2CO3
Weight: 8 Ounces (227 Grams) Purity: 99+% Pure Grade: Reagent/Food Grade Texture: Fine Powder CAS Number: 497-19-8

Sodium Carbonate [CNa2O3] 99+% ACS Grade Powder 8 Oz in a Space-Saver Bottle
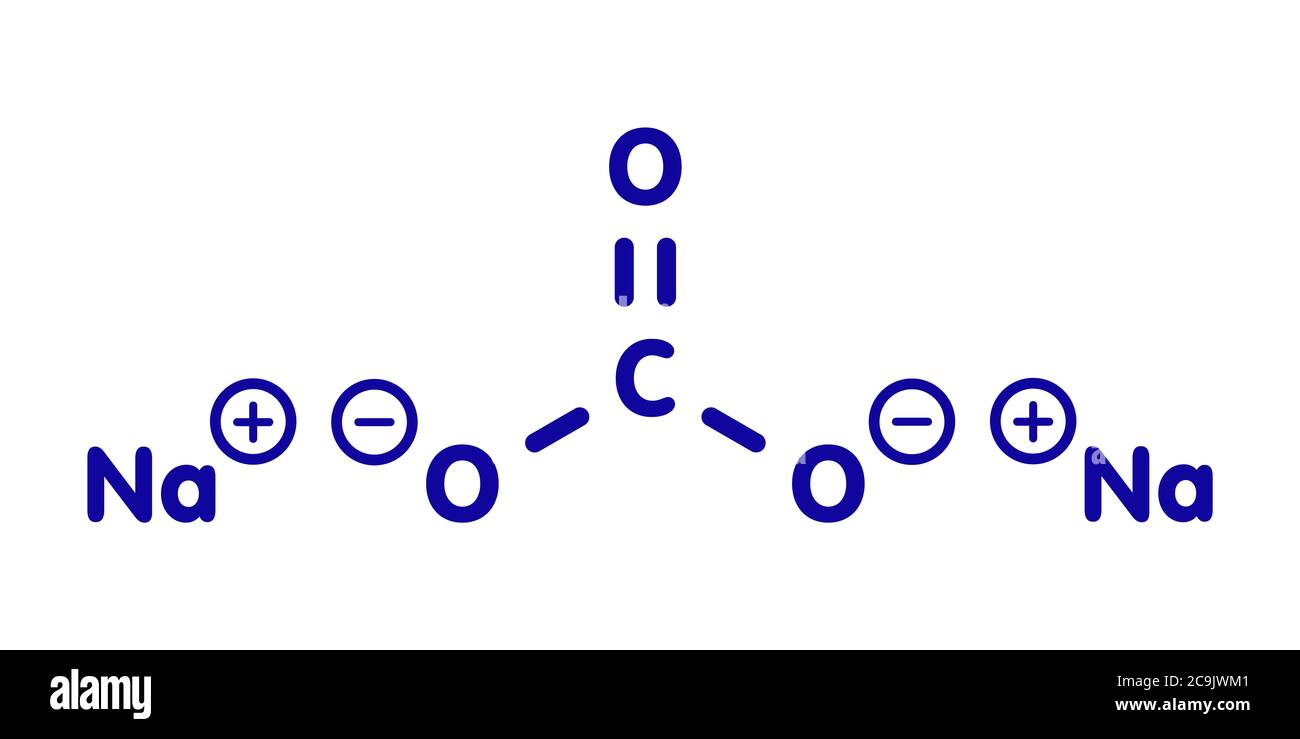
Sodium carbonate hi-res stock photography and images - Alamy
Appearance: White Solid Sodium Carbonate uses: An intermediate for the production or for the treatment of a wide variety of products in the chemicals

Sodium Carbonate (Soda Ash Dense) [Na2CO3] [CAS_497-19-8] 99.6+%, White Solid (50 Lb Bag) by Wintersun Chemical
Recommended for you
You may also like