29.8: A Catalyst Affects the Mechanism and Activation Energy - Chemistry LibreTexts
By A Mystery Man Writer
Last updated 14 Jul 2024
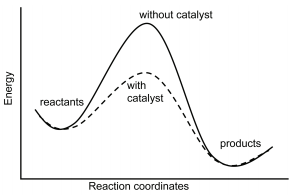
Homogeneous catalysis refers to reactions in which the catalyst is in solution with at least one of the reactants whereas heterogeneous catalysis refers to reactions in which the catalyst is present …
Homogeneous catalysis refers to reactions in which the catalyst is in solution with at least one of the reactants whereas heterogeneous catalysis refers to reactions in which the catalyst is present in a different phase, usually as a solid, than the reactants.
Homogeneous catalysis refers to reactions in which the catalyst is in solution with at least one of the reactants whereas heterogeneous catalysis refers to reactions in which the catalyst is present in a different phase, usually as a solid, than the reactants.
ADD YOUR PAGE TITLE

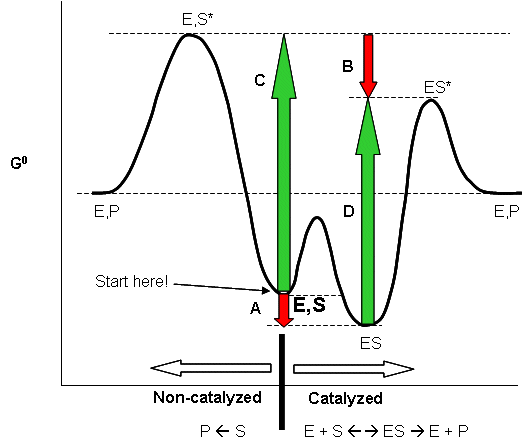
CATALYSIS

Hydrocracking of hydrotreated light cycle oil for optimizing BTEX production: a simple kinetic model

487928109-Physical-Chemistry-McQuarrie-and-Simon-Full.pdf

Chemical Kinetics: SR - 12 Class Chemistry Vol-2, PDF, Reaction Rate
4.6 Catalysis – Inorganic Chemistry for Chemical Engineers

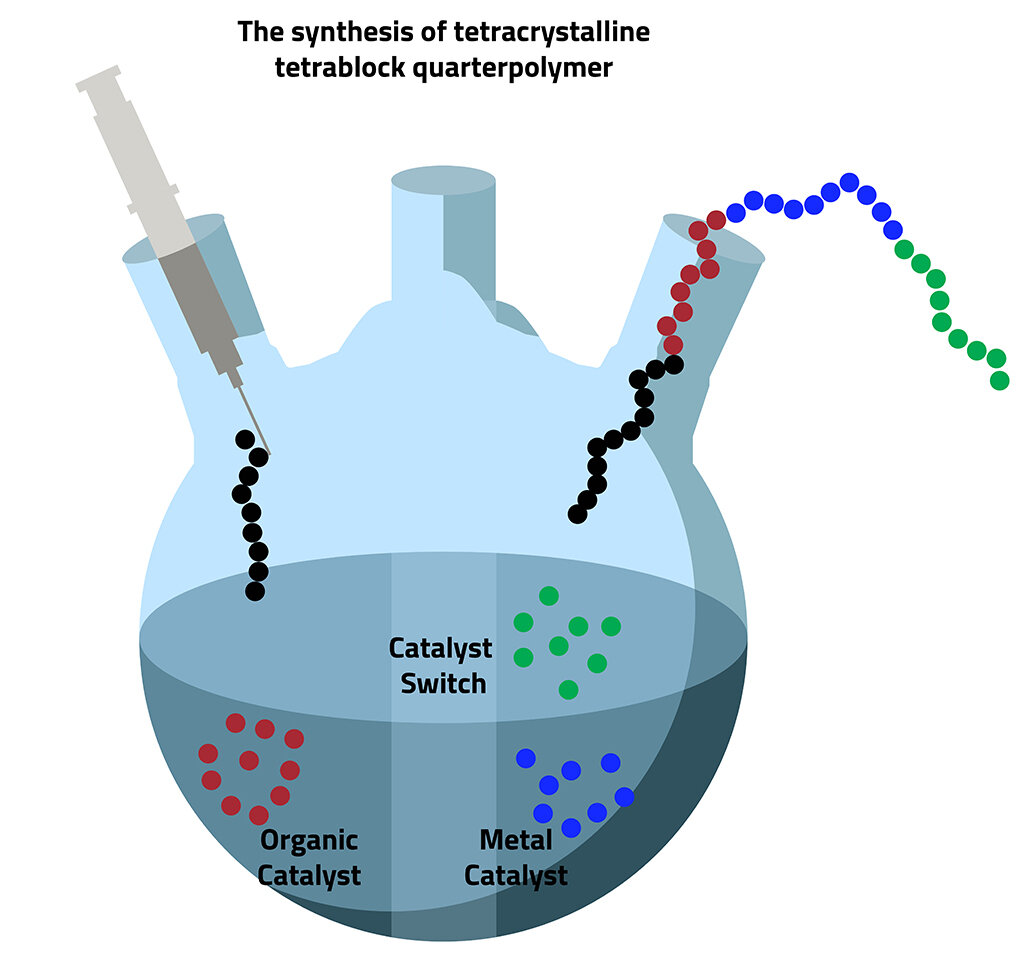
we'll always stick together, call that a forever chemical 🗣 polymers , polymerization
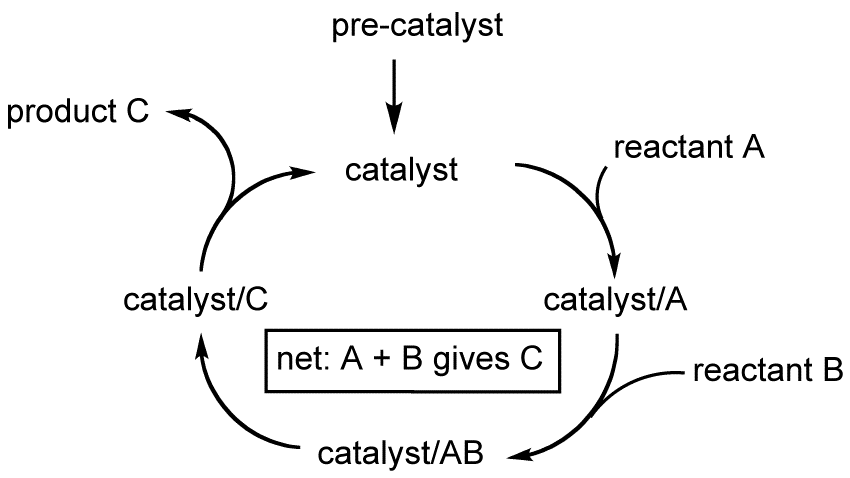
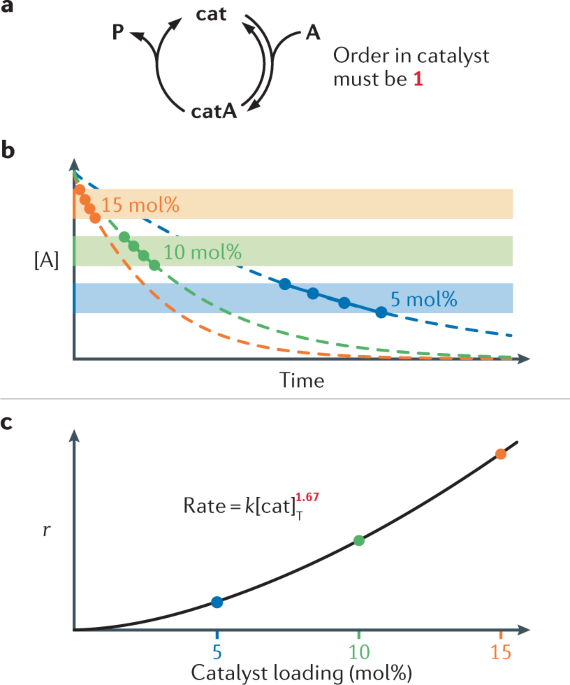
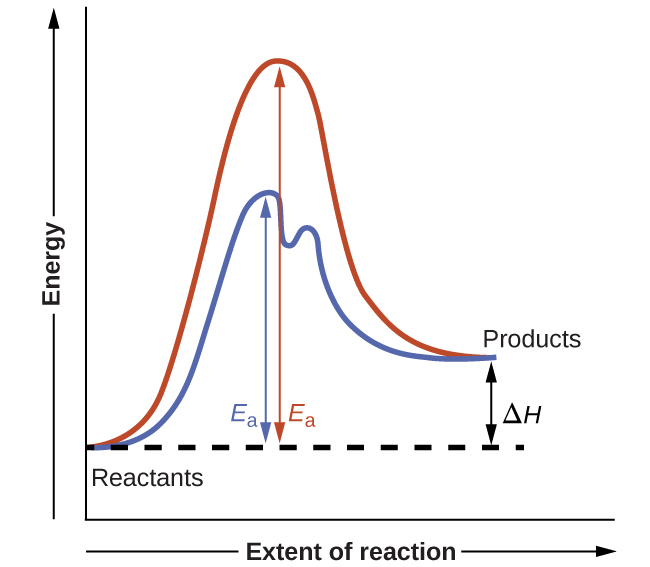
How a catalyst lowers activation energy if it introduces a reaction pathway with many Ea? What if the sum of all these Ea is greater than the Ea of the uncatalysed reaction?

Principles of Chem 2

Chemical Kinetics: SR - 12 Class Chemistry Vol-2, PDF, Reaction Rate

4.8: Catalysis - Chemistry LibreTexts

Hydrocracking of hydrotreated light cycle oil for optimizing BTEX production: a simple kinetic model
Recommended for you
You may also like