14.6: Buffers - Chemistry LibreTexts
By A Mystery Man Writer
Last updated 16 Aug 2024
A solution containing a mixture of an acid and its conjugate base, or of a base and its conjugate acid, is called a buffer solution. Unlike in the case of an acid, base, or salt solution, the …
A solution containing a mixture of an acid and its conjugate base, or of a base and its conjugate acid, is called a buffer solution. Unlike in the case of an acid, base, or salt solution, the hydronium ion concentration of a buffer solution does not change greatly when a small amount of acid or base is added to the buffer solution. The base (or acid) in the buffer reacts with the added acid (or base).
A solution containing a mixture of an acid and its conjugate base, or of a base and its conjugate acid, is called a buffer solution. Unlike in the case of an acid, base, or salt solution, the hydronium ion concentration of a buffer solution does not change greatly when a small amount of acid or base is added to the buffer solution. The base (or acid) in the buffer reacts with the added acid (or base).

569 Pages, Chapter 15.3-23.10, PDF, Aromaticity

Chapter 16.6: Buffers - Chemistry LibreTexts
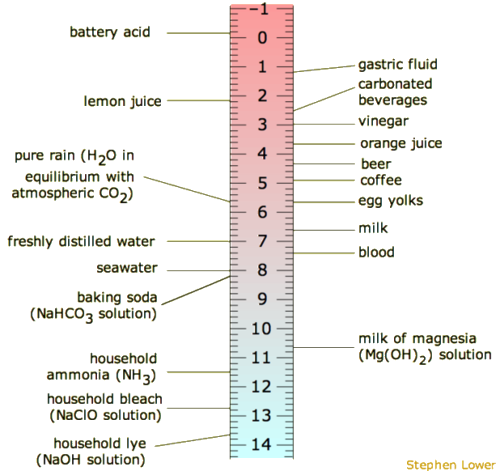
2.2: pka and pH - Chemistry LibreTexts

Direct Immersion–Solid-Phase Microextraction Coupled to Gas

Buffers - Chemistry LibreTexts, PDF, Buffer Solution
4.E: Buffer, Solubility, Common Ion Effects, and Acid-Base

chem report 3.docx - PREPARATION AND PROPERTIES OF BUFFERS Priya
Introduction to Chemistry - Chemistry LibreTexts

5.1: Day 36- Buffer Solutions - Chemistry LibreTexts

Evans PKa Table, PDF, Acid Dissociation Constant

Buffers - Chemistry LibreTexts, PDF, Buffer Solution
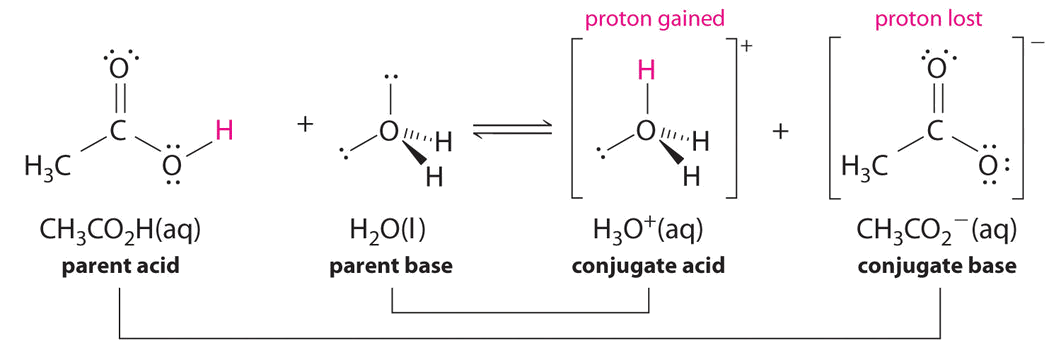
14.4: Molecular Definitions of Acids and Bases - Chemistry LibreTexts
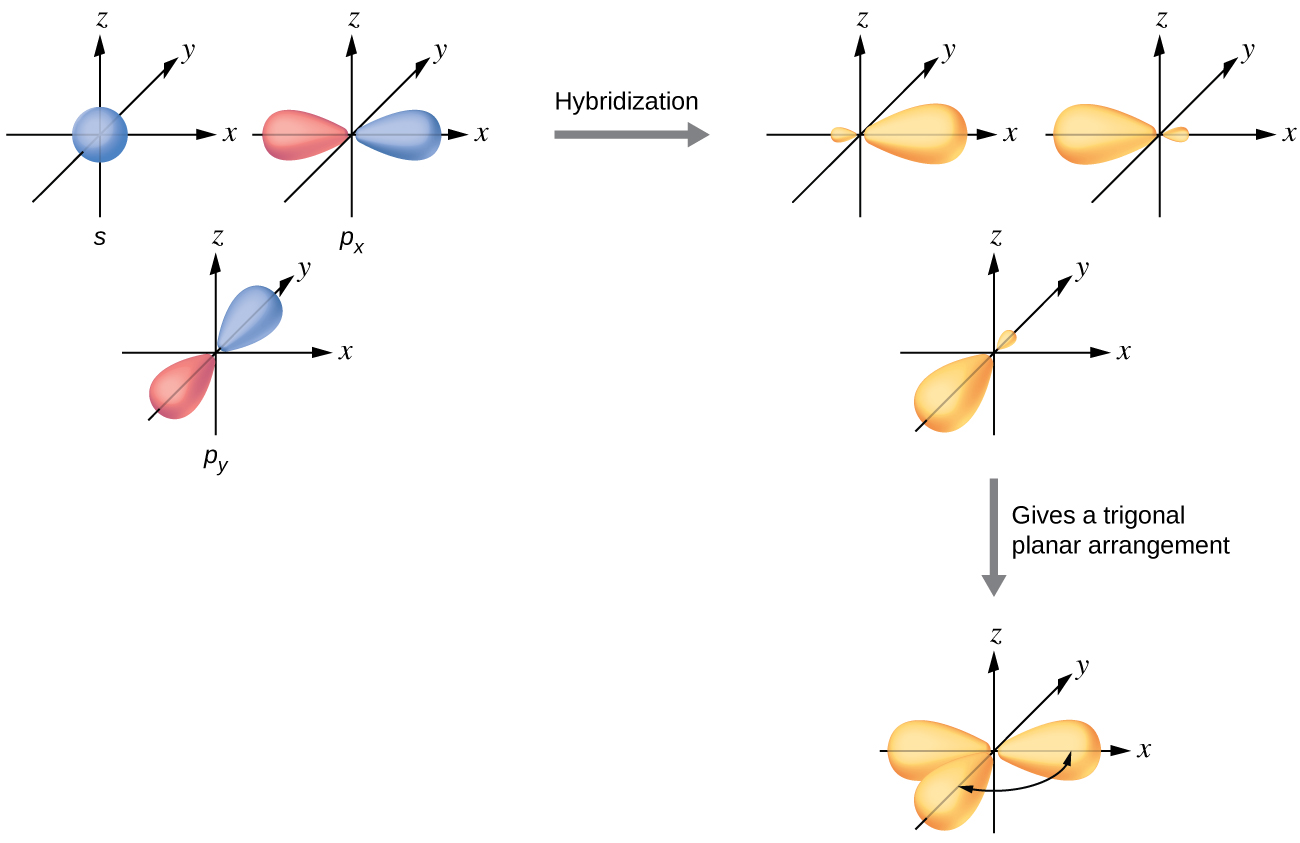
8.2 Hybrid Atomic Orbitals – Chemistry
14.6: Buffers - Chemistry LibreTexts

chem report 3.docx - PREPARATION AND PROPERTIES OF BUFFERS Priya
Recommended for you
You may also like


![Buffer Review [2024] - Is The Free Social Media Tool Plan Enough?](https://dt2sdf0db8zob.cloudfront.net/wp-content/uploads/2018/01/buffer-ease-of-use3-1.png)